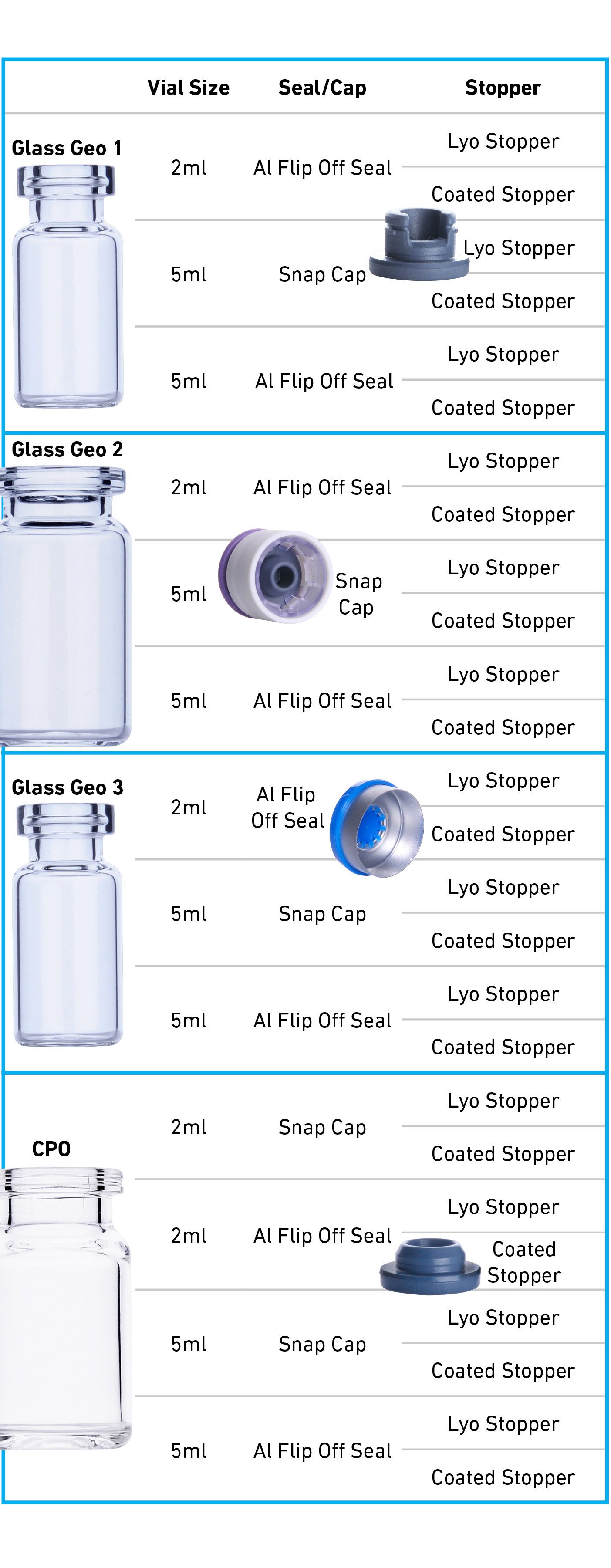
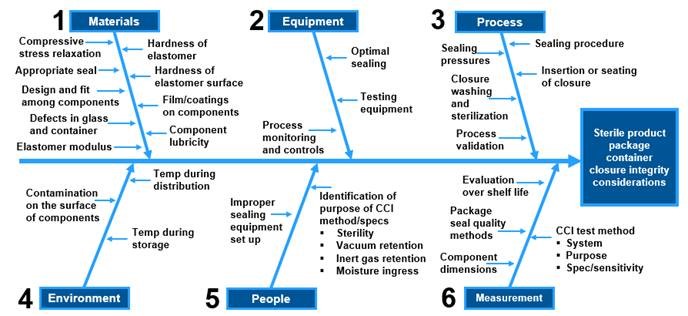
Comparative Study to Understand Compatibility of Daikyo COP vial CCS with Glass in Packaging Radiopharmaceuticals

Visual Inspection of Parenteral Products Part 5: Container Closure Integrity - From Regulations to...

Selection of a Primary Container Closure System Capable of Maintaining Closure Integrity during Cold Storage at -80°C

West Pharmaceutical Services, Inc.: Selection of a Primary Container Closure System Capable of Maintaining Closure Integrity during Cold Storage at -80°C (webinar)

Understanding Container Closure Integrity Testing | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology








/cci-pharma.jpeg?width=580&height=390&name=cci-pharma.jpeg)